The EUROIMMUN EUROArray system combines the advantages of the polymerase chain reaction (PCR) and microarray technologies for the detection of genetic markers and direct detection of pathogens. The EUROArrays are based on BIOCHIP technology with specific DNA probes. Each BIOCHIP consists of 72 DNA spots and allows duplicate determinations of up to 36 different DNA sequences, including controls. Each EUROArray slide contains five test fields, enabling up to five samples to be analyzed in parallel. Several BIOCHIPs may be coated on one field in order to increase the number of detectable DNA sequences.
Technology
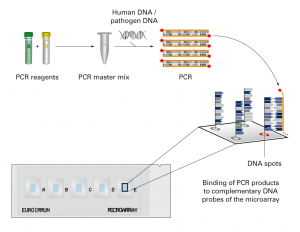
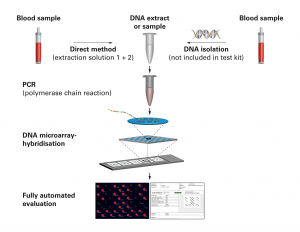 For some parameters, EUROIMMUN offers a direct procedure in which the DNA does not need to be isolated from the sample beforehand. The blood is incubated with the extraction solutions included in the kit and can then be used directly in the PCR. With all other parameters, the DNA is obtained conventionally, using a DNA isolation kit. All PCR reagents supplied in EUROArray kits are ready to use, including the DNA polymerase and the validated specific primers. The ready to use PCR reagents are just combined, and the DNA is then added to this master mix. Using PCR, the specific genetic sequences of the DNA are amplified and the PCR products labelled with a fluorescence dye during their formation. In the hybridization (incubation) of the PCR products with the EUROArray, these bind to the complementary probes of the EUROArray. The DNA microarray hybridization is performed under exact, standardized conditions using the proven TITERPLANE technology.
For some parameters, EUROIMMUN offers a direct procedure in which the DNA does not need to be isolated from the sample beforehand. The blood is incubated with the extraction solutions included in the kit and can then be used directly in the PCR. With all other parameters, the DNA is obtained conventionally, using a DNA isolation kit. All PCR reagents supplied in EUROArray kits are ready to use, including the DNA polymerase and the validated specific primers. The ready to use PCR reagents are just combined, and the DNA is then added to this master mix. Using PCR, the specific genetic sequences of the DNA are amplified and the PCR products labelled with a fluorescence dye during their formation. In the hybridization (incubation) of the PCR products with the EUROArray, these bind to the complementary probes of the EUROArray. The DNA microarray hybridization is performed under exact, standardized conditions using the proven TITERPLANE technology.
Direct procedure
For the majority of human genetic parameters, EUROIMMUN offers the Direct procedure, in which DNA needs not be isolated from the sample beforehand. The blood is incubated with the extraction solutions supplied in the kit and can then be used directly for the PCR.
With all other parameters, the DNA is retrieved using a conventional DNA isolation kit.
